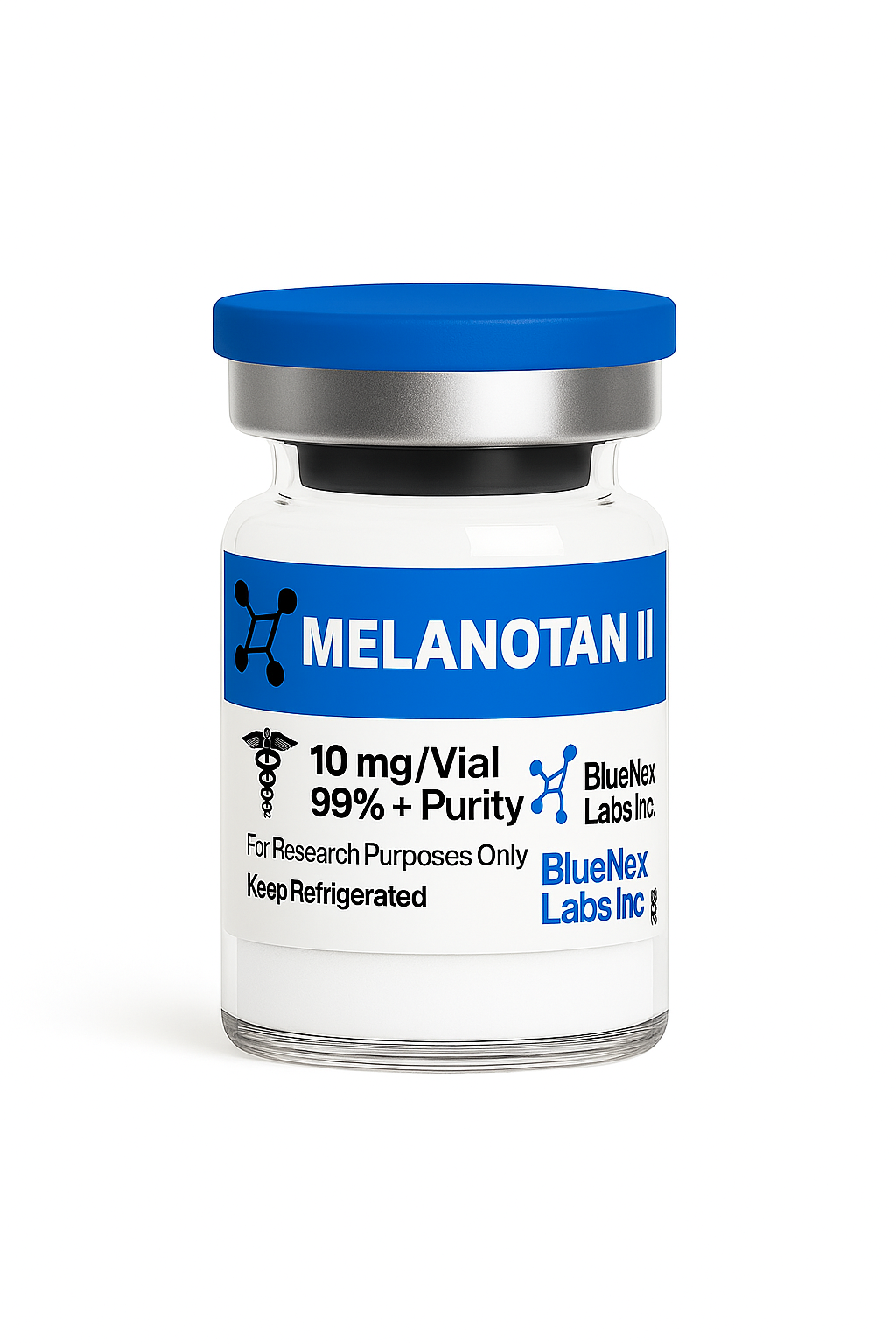
Melanotan II 10mg - COA Certified - Research Grade Peptide
Melanotan II Peptide | High‑Purity Melanocortin Analog for Receptor Research (Canada)
Melanotan 2 (MT‑2) is a synthetic analog of the body’s natural melanocyte‑stimulating hormone (MSH). By activating melanocortin receptors, it promotes increased melanin production, leading to a deeper tanning effect even with limited sun exposure.
In addition to its role in pigmentation, Melanotan 2 has been explored for its potential influence on appetite regulation and sexual function, due to its interaction with specific neural pathways involved in these processes.
Key Research Areas & Mechanistic Highlights
🎨 Pigmentation & Melanin Production
• Synthetic analog of endogenous α‑MSH
• Activates MC1R receptors on melanocytes
• Studied for its ability to increase eumelanin synthesis
• Used in research exploring UV‑independent pigmentation pathways
🧠 Neural Pathways & Receptor Activity
• Interacts with multiple melanocortin receptors (MC1R–MC5R)
• Examined for its influence on CNS signaling networks
• Studied in relation to neuroendocrine communication and behavioral pathways
🍽️ Appetite & Energy Regulation
• Investigated for potential effects on satiety‑related signaling
• Explored in metabolic research involving melanocortin‑mediated pathways
• Used to study mechanisms related to energy balance and feeding behavior
🔥 Sexual Function & Arousal Pathways
• Preclinical interest due to interactions with MC3R and MC4R
• Studied for its role in neural circuits associated with sexual response
• Considered a model compound for understanding melanocortin‑linked arousal mechanisms.
🔬 Research Applications (Non‑Medical)
Melanotan II is widely used in melanocortin‑receptor research, particularly involving MC1R, MC3R, MC4R, and MC5R signaling in controlled in‑vitro systems.
Key research applications include:
Receptor‑binding assays — used to evaluate affinity and receptor‑subtype selectivity across the melanocortin family.
Intracellular signaling studies — supports research into cAMP accumulation, PKA activation, and downstream transcriptional responses.
Structure–activity analysis — applied in comparisons of cyclic vs. linear melanocortin analogs to evaluate stability and receptor bias.
Peptide‑stability modeling — used to study enzymatic degradation and cyclic‑peptide resistance to proteolysis.
Analytical method development — suitable for validating chromatographic separation of cyclic heptapeptides.
🧬 Molecular Characteristics
Type: Cyclic heptapeptide melanocortin analog.
Form: Lyophilized powder (10mg).
Identity: Verified via MS and HPLC/UPLC.
Biochemical Notes:
Cyclic structure enhances stability and receptor affinity.
Solubility varies with pH; performs best in mildly acidic buffers.
Stability:
Stable in dry form; sensitive to hydrolysis and oxidation in solution.
Avoid prolonged exposure to elevated temperatures.
❄️ Storage & Handling
Store at –20°C, protected from humidity and light.
Reconstituted solutions should be stored at 2–8°C and used promptly.
Avoid repeated freeze–thaw cycles.
📄 Documentation / COA Visibility
COA includes MS identity, HPLC purity, solvent system, and lot traceability.
RUO‑only; not for human or veterinary use.
⚠️ Compliance Notice
Melanotan II is supplied for laboratory research only. Not for human or veterinary use. No therapeutic, metabolic, or performance‑related claims are made or implied.
Melanotan II Peptide | High‑Purity Melanocortin Analog for Receptor Research (Canada)
Melanotan 2 (MT‑2) is a synthetic analog of the body’s natural melanocyte‑stimulating hormone (MSH). By activating melanocortin receptors, it promotes increased melanin production, leading to a deeper tanning effect even with limited sun exposure.
In addition to its role in pigmentation, Melanotan 2 has been explored for its potential influence on appetite regulation and sexual function, due to its interaction with specific neural pathways involved in these processes.
Key Research Areas & Mechanistic Highlights
🎨 Pigmentation & Melanin Production
• Synthetic analog of endogenous α‑MSH
• Activates MC1R receptors on melanocytes
• Studied for its ability to increase eumelanin synthesis
• Used in research exploring UV‑independent pigmentation pathways
🧠 Neural Pathways & Receptor Activity
• Interacts with multiple melanocortin receptors (MC1R–MC5R)
• Examined for its influence on CNS signaling networks
• Studied in relation to neuroendocrine communication and behavioral pathways
🍽️ Appetite & Energy Regulation
• Investigated for potential effects on satiety‑related signaling
• Explored in metabolic research involving melanocortin‑mediated pathways
• Used to study mechanisms related to energy balance and feeding behavior
🔥 Sexual Function & Arousal Pathways
• Preclinical interest due to interactions with MC3R and MC4R
• Studied for its role in neural circuits associated with sexual response
• Considered a model compound for understanding melanocortin‑linked arousal mechanisms.
🔬 Research Applications (Non‑Medical)
Melanotan II is widely used in melanocortin‑receptor research, particularly involving MC1R, MC3R, MC4R, and MC5R signaling in controlled in‑vitro systems.
Key research applications include:
Receptor‑binding assays — used to evaluate affinity and receptor‑subtype selectivity across the melanocortin family.
Intracellular signaling studies — supports research into cAMP accumulation, PKA activation, and downstream transcriptional responses.
Structure–activity analysis — applied in comparisons of cyclic vs. linear melanocortin analogs to evaluate stability and receptor bias.
Peptide‑stability modeling — used to study enzymatic degradation and cyclic‑peptide resistance to proteolysis.
Analytical method development — suitable for validating chromatographic separation of cyclic heptapeptides.
🧬 Molecular Characteristics
Type: Cyclic heptapeptide melanocortin analog.
Form: Lyophilized powder (10mg).
Identity: Verified via MS and HPLC/UPLC.
Biochemical Notes:
Cyclic structure enhances stability and receptor affinity.
Solubility varies with pH; performs best in mildly acidic buffers.
Stability:
Stable in dry form; sensitive to hydrolysis and oxidation in solution.
Avoid prolonged exposure to elevated temperatures.
❄️ Storage & Handling
Store at –20°C, protected from humidity and light.
Reconstituted solutions should be stored at 2–8°C and used promptly.
Avoid repeated freeze–thaw cycles.
📄 Documentation / COA Visibility
COA includes MS identity, HPLC purity, solvent system, and lot traceability.
RUO‑only; not for human or veterinary use.
⚠️ Compliance Notice
Melanotan II is supplied for laboratory research only. Not for human or veterinary use. No therapeutic, metabolic, or performance‑related claims are made or implied.